Virginia Postrel’s The Fabric of Civilization, Part 4
Special thanks to Petra Juarez, ceramic artist and artistic innovator, for permission to use her many photos.

In the fall of 2024, San Diego’s Mingei International Museum exhibited “Indigo,” which included living indigo plants, as well as myriad contemporary and historical textiles dyed with indigo; in addition, the museum’s gift shop offered opportunities for amateurs to use indigo dye.
This is the fourth of five blogs discussing Virginia Postrel’s book on textile history:
Virginia Postrel. (2020). “Chapter Four: Dye” (pp. 109–146). The Fabric of Civilization: How Textiles Made the World. New York: Basic Books.
The first three blogs are
- https://bird-brain.org/2026/04/01/fiber/
- https://bird-brain.org/2026/04/08/thread/
- https://bird-brain.org/2026/04/15/cloth/
The fifth (and final) blog about Postrel’s book is forthcoming.
Indigo Blue
Archeologists have been studying the 14,000-year-old site of Huaca Prieta, Peru, where they found 6,000-year-old textiles dyed in blues, reds, and greens — challenging colors to produce using only natural dyes. The Huaca Prietans had no pottery — gourds served as containers — but they had sophisticated dyed textiles. With natural dyes, it’s easy to produce browns, yellows, and grays, but to get textiles dyed blue, red, or green requires extensive experimentation in dyeing techniques. “The ancient dyer . . . was an advanced empirical chemist” (p. 110).
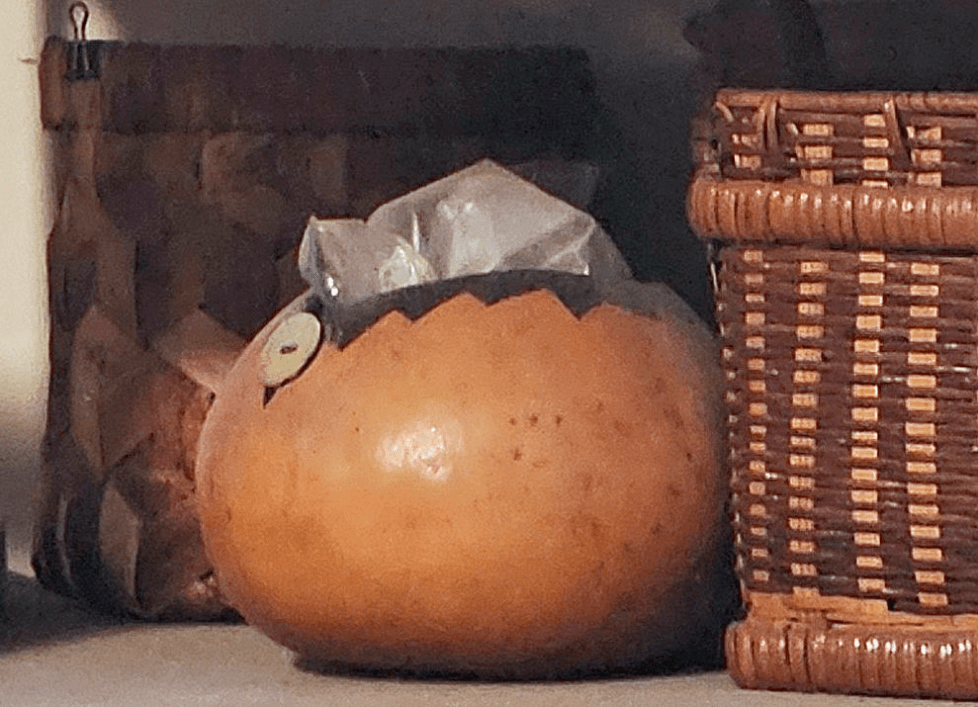

Figure 04-01. Before ceramics were being used, people used gourds and baskets for storage, but even at that time, people were dyeing and weaving textiles. (Photo of indigo-dyed textiles by ceramic artist Petra Juarez, who also explores other artistic media and shares her skills as a registered nurse.)
The blue dye used in Huaca Prieta came from the indigo plant, still in use today. Indigo contains indican, which can also be found in European woad plants and various other species; the particular indican-bearing plant in a given location depends on the climate and soil conditions of the locale. Woad is a cabbage; the Indigoferra tinctoria plant of the Americas, Africa, and much of Asia is a legume; the indigo-bearing plant of Japan is a kind of buckwheat. Numerous other wild plants contain indican, too.
Though we say the use of indigo yields a “natural dye,” that grossly understates the degree of technological invention involved in making useful dye from the wild plant. Here are some of the steps for producing indigo dye:
- 1. Harvest the leaves and soak them in water, so that the leaves’ cellular structure breaks down, releasing both the indican and an enzyme into the water. The enzyme initiates a chemical reaction that splits the indican into a sugar and molecules of indoxyl. When the highly reactive indoxyl comes into contact with the oxygen in the water (H2O), they quickly bond and form indigotin, the blue pigment of indigo.
- 2. The indigotin doesn’t dissolve in water, so it precipitates to the bottom of the container as a slurry. If you’re using the pigment for painting or drawing, you’re all set. Just remove the pigment slurry, ditch the indigo leaves, and you’re ready to go.


Figure 04-02. The Mingei International Museum’s “Indigo” exhibit showed dried indigo plants from Turkey, Japan, and Florida (U.S.A.), among many other items.
- 3. Unfortunately, the slurry of pigment won’t adhere to textiles, and it won’t dissolve in water. If you want to use indigotin in a liquid to dye textiles, you have to transform the pigment into a water-soluble dye that will bind with fabric. To get the indigotin to dissolve in water, you have to change the pH of the water to be intensely alkaline (a base). To get highly alkaline water, you add wood ashes to plain clean water.
- 4. Next, you extract the indigotin slurry from the gloppy leaves and water, and you add the indigotin slurry to the alkaline (wood-ashy) water.
- 5. By adding the indigotin slurry to the highly alkaline water, the indigotin reacts to the alkaline water by forming a water-soluble compound, leuco-indigo. So far, so good.
- 6. But the leuco-indigo wants to bind with oxygen again and revert back to being indigotin. Yikes! Somehow, the water’s oxygen levels must be reduced. One way to remove the oxygen in the water is to ferment it, using bacteria; dyers around the world have added assorted foodstuffs (e.g., dates, bran, honey, rice) to the water, to feed bacteria to ferment the concoction. They hadn’t been reading textbooks about microbiology, chemistry, or fermentation; they learned which foods to add solely through trial-and-error experimentation.
- 7. Later experimental chemists — that is, dyers — discovered that they could reduce the oxygen levels — while not knowing that oxygen existed — by adding iron compounds to the dye vat. The iron compounds oxidized and precipitated out of the dye solution as rust.


Figure 04-03. Ceramic artist Petra Juarez is dyeing cotton fabric, using prepared indigo.
- 8. Once the leuco-indigo dissolves, bubbles form at the top of the dye vat, signaling the dyer to immerse the fabric to be dyed. In the dye vat, the leuco-indigo penetrates the fibers of the fabric and turns them greenish. The dyer then removes the fabric, exposing it to the air, where the leuco-indigo binds with oxygen, becoming indigotin again, and turning the fabric indigo blue. To intensify the color, the dyer repeats the dipping and removing of the fabric. The more dips, the darker the color, as layers of indigotin build up in the fabric.
What if a dyer doesn’t have time to do all these steps between dawn and dusk? Luckily, the dyer can remove the indigotin slurry and set it aside. The slurry can storing be stored wet, as damp paste or as moist balls, wrapped in indigo leaves. For longer-term storage, it can be dried into lightweight, portable cakes. Indigo dyes stick not just to silk or wool, but also to plant-based fibers such as cotton or flax (linen). Indigo-dyed fabric doesn’t fade in sunlight or rinse out when washed — staying colorful for thousands of years! — though it can eventually be rubbed off with wear (e.g., your blue jeans).
One major drawback, though: Indigo dyeing stinks. Even dyeing with woad is so stinky that Queen Elizabeth I forbade English dyers from indigo dyeing within 8 miles of any of her palatial homes.



Figure 04-04. Before her wedding, ceramic artist Petra Juarez used prepared indigo dye to dye all the textiles for her wedding reception, from tablecloths to napkins. Luckily, contemporary prepared indigo does not stink, so she and her collaborators didn’t have to tolerate a stench, and her beautifully dyed textiles smelled fresh and clean.
Tyrian Purple
Dyes get even stinkier when we get to purple dyes made from a marine snail. The ancient Phoenician city of Tyre (by 1500 B.C.E.) is said to have discovered the snail’s purple dye when a noble’s dog (or perhaps a deity’s dog) bit down on a sea snail, turning the dog’s mouth purple. The Phoenicians actually extracted “Tyrian purple” dye from three different murex marine snails: Bolinus brandaris and Stramonita hemastoma, which produced a red-violet dye; and Hexaplex trunculus, which could be used for dying blue, blue-violet, or red-violet hues.
Dyers who made and used Tyrian purple had no college degrees in chemistry or marine biology, so how did they know how to make the dye? Once again, trial-and-error experimentation. The earliest records of how this dye was produced come from the Roman naturalist Pliny the Elder, in his Natural History encyclopedia (A.D. 77–79). His details about the snails and the resulting dyes may have been meaningful to the dyers of his time, but the significance has been lost since then.
The living snails have a gland containing the colorless indoxyl compound, which the snails release when they die. To ensure that the indoxyl wasn’t released into the sea, dyers kept aquaria filled with these snails. When ready to use the snails for dye, the dyers opened the snail shells and extracted the glands. If a snail was too small to surgically remove the gland, it was crushed. The dyers put “the glands, secretions, and snails into a vat of water kept warm, but not boiling,” then they added salt to the mixture.

Figure 04-05. These snail shells are extremely hard. Wikipedia describes this photo as “purple dyed fabric with their corresponding sea snail: Bolinus brandaris, Hexaplex trunculus, Stramonita haemastoma (Thais haemastoma).”
From an exhibit of the Museum of Natural History in Vienna, 29 November 2018; file, Purple Purpur.jpg. The copyright holder of this work licensed publication under the Creative Commons Attribution-Share Alike 4.0 International license. You are free: to share – to copy, distribute and transmit the work, to remix – to adapt the work, under the following conditions: attribution – You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use. share alike – If you remix, transform, or build upon the material, you must distribute your contributions under the same or compatible license as the original.
After the mixture steeped for three days (STINKY!), it was transferred to a metal cauldron, where more water was added, and the residual snail solids were removed. The resulting solution was cooked for an additional nine days (more STINK!), then a fleece was dipped into the solution to test whether the dye was ready. Pliny described the resulting color as “the color of coagulated blood: dark when observed from the front, with bright reflections when seen from an angle” (p. 118). Perhaps to ancient Tyrians, this color was highly attractive, but it definitely wouldn’t compete with modern-day synthetic purples. And one more thing: Even the dyed and washed Tyrian purple fabric . . . stank.
Perhaps to our eyes — and noses! — Tyrian purple fabric wouldn’t be worth the trouble. But to ancient Tyrians, it was the ultimate symbol of status, wealth, opulence. It was definitely a lot of trouble to make. A modern-day archeologist, Deborah Ruscillo, decided to replicate, as much as possible, the traditional Tyrian purple dye. First, she gathered about 100 snails/hour, filling a bucket of seawater with 800 snails in 8 hours. Then she had to pry open each individual tough snail shell, which turned out to require two steps/snail: Pound an awl into the main snail-shell’s whorl to poke a hole, then use the hole to pry open the shell.



Figure 04-06. Not every experiment in natural dyes is a raging success. Years ago, I had unwittingly stained a couple of textiles and decided to see what would happen if I tried to dye them with beet skins. After all, drips of beet juice had “dyed” fabric previously, so it should work, right? I boiled up a pot of beet skins, removed the skins from the pot, added the cloth, and boiled the fabrics in the beet juices. It looked great when I set them out to dry. . . . Not so great after they had dried in the sun, however. I still use them as towels, though.
Next, Ruscillo and an assistant extracted the gland from each snail and discarded the rest, along with all the snail shells. They put the accumulating glands into a pot of water with a tight-fitting lid, covering an increasingly slimy concoction. Meanwhile, flies snuck into the pot and laid their eggs inside whenever the lid was lifted. To get rid of the maggots, while not ruining the dye, they brought the pot’s temperature up to just below the boiling point. (Remember, all the while, it stinks!) Throughout this process, their hands were being dyed purple, which no amount of washing or cleansing could remove — and it stank!
Once Ruscillo had the dye, she tried it with four different fabrics — wool, cotton, nubby raw silk, and smooth satin-weave silk. She also used five different types of dye baths with the snail stew: seawater alone, freshwater alone, seawater with urine (probably didn’t help with the smell!), seawater with alum (potassium aluminum sulfate, a mineral salt often used for dyeing), and seawater with vinegar. That yielded 4 × 5, 20 different tests. She also modified Pliny’s instructions slightly. Instead of the 9-day cooking step, she steeped the mixture for 3 days at 176 degrees F. (80 degrees C.; 212 F. is boiling, 100 degrees C.). After 3 days, she removed the snail glands and dunked the fabric samples. She also tried dunking fabric into the seawater mixture, with no steeping time.
Of all the fabrics, wool absorbed the dye the best — along with the stench, which continues to permeate the fabric, despite repeated washings, almost 20 years later. Both silk fabrics also absorbed the dye better than did the cotton. The only differences she noticed among the various dye baths were that the freshwater dye bath was less colorfast and faded over time, and the urine-seawater dye bath seemed a bit more vibrantly purple.
The addition of salt, suggested by Pliny, may have helped keep the snail’s flesh from rotting as quickly. Seawater itself has a pH of 8.3, so it’s already slightly alkaline (neutral pH being 7), so maybe ancient dyers didn’t need to add alkali to seawater. Ruscillo also discovered that the un-steeped seawater mixture produced fabric with a beautiful blue, reminiscent of the biblical blue fringe on Jewish men’s talit ritual prayer cloths.
Ruscillo found that it would take a few hundred snails to dye enough fabric to make trim or a small lightweight garment, but for a large wool cloak, many thousands of snails would need to be harvested and processed. For the snail harvesting alone, hundreds of hours would be needed. And, of course, there’s the stench.



Figure 04-07. When using natural dyes, an artisan can make approximate guesses about how the dyed fabric will look after it has dried, but results are often unpredictable; naturally dyed textiles aren’t as uniform as other textiles. The silk (and cotton) textiles shown here were all dyed by ceramic artist Petra Juarez, using an array of natural dyes and mordants, as well as special techniques to create intriguing patterns. Petra enjoys exploring — and mastering — many other artistic media, as well as her day job as a registered nurse.
Ruscillo likened the ancient dyeing process to cooking: Through trial and error, an experienced dyer would develop some recipes for how to make useful dyes. The dyers wouldn’t understand the underlying chemistry, but they tweaked the recipes until they obtained the desired results. (For more on Tyrian purple, see https://en.wikipedia.org/wiki/Tyrian_purple .)
Red Dyes
By the time of the Italian Renaissance, red dyes had replaced Tyrian purple dyes as signs of wealth and power (perhaps Italians of the Renaissance were more averse to stinky purple dye). In 1548, after spending 16 years learning from artisanal dyers, Gioanventura Rosetti published The Plictho, the first known manual for professional dyers. In his manual, 35 of the recipes were formulas for red dyes, and 21 were for black dyes. (More than a century later, in 1677, Marx Ziegler did for weavers what Rosetti did for dyers, making public former trade secrets.)
Among the red dye recipes noted by Rosetti, dyer’s madder, Rubia tinctorum, had been used for dyeing fabrics since as long ago as A.D. 73, found in the garments of Jewish rebels at Masada, with colors from bright red to salmon pink to deep burgundy to purple, and more. How can one plant yield so many variations? First, the madder’s root itself contains two different colorant chemicals: an orange-reddish alizarin, and a purplish-hued purpurin. The proportions of each color in a given plant root will vary, according to the plant subspecies, the soil condition, and even the age of the plant when the root is harvested. A knowledgeable, skilled dyer could use these variations to produce differing dye colors.
Dyers experimented not only with the botanical dyeing agents, but also with additives to the water, which could change both the color of the dye and the color-fastness of the dye. For instance, fermented bran water gave madder a bluish tone (and made it stink!), and white tartar (left over from wine fermentation) added an orange hue to madder’s color.
Dyers also learned to use mordants—additives that bound the dye to the fabric, so it wouldn’t wash out or otherwise fade over time. Mordants were mostly metal salts, such as alum. The mordant would bind with the fiber, binding the dye to the fiber at the same time. Some mordants also altered the shades or even the colors. Iron compounds usually darkened or even dulled the color.
According to the Oxford English Dictionary, mordant meant the tongue of a buckle, which held the buckle from undoing or sliding (ca. 1200); it also meant “biting” from mordre, “to bite.” Both meanings apply to how a mordant can make a dye bite or poke into a fiber or fabric.

Figure 04-08. Though few contemporary dyers would go to the extreme lengths of Deborah Ruscillo, many dyers will experiment with using different natural dye mediums and different mordants (to get the dye to adhere to the fabric). This photo documents the mordant experiments of ceramic artist Petra Juarez, using cotton fabric. Top row, no mordants were used; left fabric swatches used avocado peels and pits, right used onion skins. Middle row, iron used as a mordant (left, avocado; right, onion). Bottom row, alum was the mordant.
In addition to madder, dyers also used brazilwood and grana to make red dyes. Brazilwood was extracted from the hardwood of some trees and had become relatively abundant by the time Rosetti wrote his manual. On its own, brazilwood can fade to a dull color over time, but it can add depth to other colors when used in combination.
The substance grana (also called “kermes”) was obtained by harvesting thousands of tiny parasitic insects living wild on European oak trees, then grinding them up to extract the dye. It was quite expensive, but it was loaded with more color than the madder or the brazilwood, so it continued to be used.
Meanwhile, in the New World, for centuries, Mexican dyers had been cultivating parasitic cochineal insects on the nopal cactus. Mexican spinners and weavers were using cochineal to produce intensely red fibers and textiles for the Aztec rulers who continually demanded more.
Soon after Spaniards arrived in the New World, they exploited anything they could find to enrich the Spanish rulers. Pretty soon, they lit upon the cochineal dyes, and by the 1550s, European traders were importing cochineal insects, along with many other products from the New World.

Figure 04-09. These parasitic cochineal insects are being cultivated on cactus, for later harvesting, to use for dyeing.
The photo, “Cochineal on opuntia, 7 July 2022,” by Jengod, is from Wikipedia, https://en.wikipedia.org/wiki/Cochineal . This file is licensed under the Creative Commons Attribution-Share Alike 4.0 International license. You are free: to share – to copy, distribute and transmit the work; to remix – to adapt the work, under the following conditions: attribution – You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use. share alike – If you remix, transform, or build upon the material, you must distribute your contributions under the same or compatible license as the original.
At first, grana and cochineal were used interchangeably, but cochineal dye contains 10 times more colorant than grana, and it is much more colorfast. European demand for cochineal grew; by 1600, cochineal was essential to European dyers. In Mexico, it became increasingly lucrative for peasant farmers to grow nopal and its cochineal parasites for the European market. Many peasant farmers turned their land from food crops to cochineal/nopal, buying food from local markets and driving up the prices of food — much to the consternation of the local elites who had previously enjoyed being able to dominate the local peasantry. Spain zealously tried to control its monopoly on the cochineal trade, while Queen Elizabeth I directed her favorite pirates to target cochineal-bearing ships on their escapades.
In Europe, dyers had found it challenging to get linen, cotton, or other plant-based cellulose fibers to absorb and hold vivid colors. These fibers resisted absorbing most dyes and quickly released any dyes into the wash. To get vividly colorful cottons and linens, Europeans had to weave in additional threads or embroider the fabric, using fibers from animal sources.
Meanwhile, in India, spinners, dyers, and weavers were producing lightweight, colorful cotton cloth. Their cotton fabric was itself a wonder — as breathable, lightweight, and washable as linen, as affordable as wool, but not as scratchy or stiff as wool or linen, and not as costly as silk. It was soft, lightweight, and vividly colorful. What’s more, by the time Europeans were aware of Indian cotton fabric, Indians had mastered techniques for dyeing their fabrics a rainbow of colors. Not only could they produce an array of colors overall, but they could produce vibrant complex designs, motifs, and prints on a single textile.
Synthetic Dyes
Once Europeans saw these marvels of textile design, they couldn’t get enough of them. Which posed a big problem for European textile workers, especially dyers. At first, European politicians responded with severe regulations, which failed to achieve the desired results. At last, in the early 1700s, governments started to support research to develop the most effective dyes, at about the same time that the science of chemistry was burgeoning. In 1791, Claude Louis Berthollet published a book applying the newfound knowledge of chemistry to fabric dyes. His book left much to be desired in the application of science, but it was a good start in understanding why some dye practices worked and some didn’t.
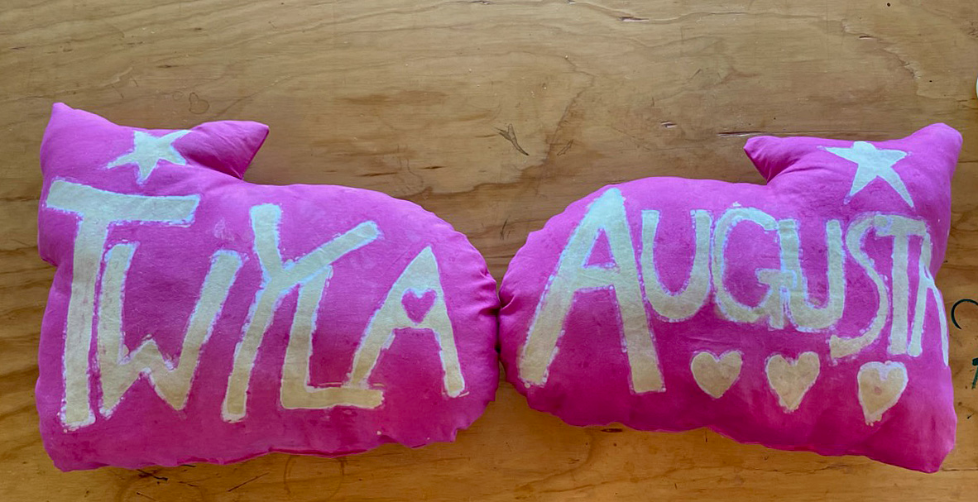

Figure 04-10. More than 2,000 years ago, to dye textiles with intense colors, the process was not only time consuming and difficult, but also stinky, but nowadays, synthetic dyes allow enthusiasts to dye fabrics in almost any colors imaginable, relatively quickly and easily. (Photo of synthetic-dyed pillows by ceramic artist and multitalented artisan Petra Juarez.)
Up until the mid-1800s, dyers relied on natural dyes, which they had learned to tweak and modify to produce an array of colors. But it wasn’t easy to decide at the outset to create a particular color of dye and be absolutely sure that the resulting dye would indeed be that color. And some colors — such as many greens — were nearly impossible to achieve with any regularity or predictability.
In the 1850s, August Kekulé revolutionized chemistry by showing that chemicals had a molecular structure, deepening the understanding of chemicals. About that time, August Wilhelm Hofmann discovered that petrochemicals (made from coal tar, oil, and other hydrocarbons) could produce aniline chemicals similar to such plant products as dyes and medications. The possibilities seemed promising, partly because coal tar was an industrial waste product remaining after purifying coal for use as a fuel. If coal tar could be used productively, it could yield greater control over manufactured products, compared with natural plant products.
One of Hofmann’s students, William Perkin, decided to try to make a medication — quinine, used to treat malaria — but sadly, his various experiments failed to do so. One of his experiments, however, left an odd-looking precipitate, which he dissolved in some denatured alcohol — and it turned a stunning purple. At the time of Perkin’s discovery, the textile industry was booming, prompting a big desire for usable dyes. Perkin tried using the purple solution to dye some silk fabric, with great success.
Perkin left college to pursue a business in manufacturing what he called “Tyrian purple” dye, but it was extremely challenging to switch from producing small amounts of dye to producing commercial volumes of dye. Also, though Tyrian purple dye worked beautifully on silk fibers, cotton fibers resisted absorbing it, and affordable cotton was far more popular than costly silk. Once techniques were devised to commercially produce Perkin’s dye in cotton-friendly form, it became popularly known as “mauve.” Soon, other chemists were producing other shades of aniline purples, including Hofmann, Perkin’s old teacher. By the end of the 1800s, chemists were also making red and blue dyes, replacing madder and indigo. Fields of madder, indigo, and other dye-crop plants were turned to other purposes (e.g., to vineyards in France).

Figure 04-11. The Mingei International Museum’s “Indigo” exhibit mostly focused on historical uses of indigo for dyeing various textiles, but the exhibit also showed numerous contemporary textile artists who are using indigo dyes, rather than synthetics.
Postrel reminds readers that though petrochemicals clearly contribute to pollution and environmental hazards, plant dyes are far from benign. Dyers need huge quantities of water to dye fibers or fabric: soak, rinse, dump, soak, rinse, dump, repeat, . . . . Fuel must be used to heat the water and the dye solutions; and the actual dye compounds can be stinky — urine, foul-smelling indigo, the vomit-like stench of bran water, the rotting-flesh odor of snails, and more.
On the other hand, Postrel points to an ultramodern textile company, Swisstex (https://www.swisstex-ca.com/Swisstex_Ca/Welcome.html ), located in Los Angeles, California, which custom-dyes “organic cotton, recycled cotton, recycled polyester, recycled nylon, biodegradable synthetics” (from its website, swisstexdirect.com), using natural gas enhanced by the recycling of hydrocarbons it has broken down, and transforming its emissions into carbon dioxide and steam. According to Postrel, they “use less water per pound [of fabric] than any dye house on [Earth]” (p. 145). They also have made myriad improvements to cut the time needed for dyeing dark colors from 12 hours to 4–5, which also saves on fuel and on carbon emissions.


Figure 04-12. These images were taken from these Swisstex website pages, https://www.swisstex-ca.com/Swisstex_Ca/Laboratory.html and https://www.swisstex-ca.com/Swisstex_Ca/Environment.html .
Coming soon (before the end of April): Part 5, my final blog about Postrel’s book.
Text by Shari Dorantes Hatch.
Image sources indicated in captions; images lacking sources are by Shari Dorantes Hatch.
Copyright © 2026. All rights reserved.

Leave a comment